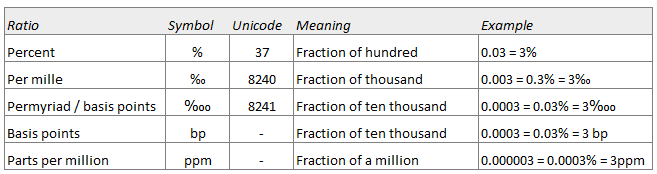
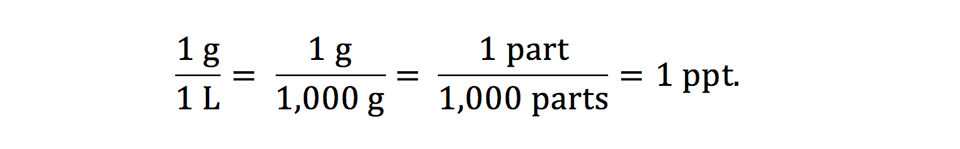SOLVED: Percent by definition is parts per hundred: % (wlw) (mass of solutelmass of solution) x 100. For very dilute solutions, chemists will often use parts per thousand or parts per million (

SOLVED: Percent by definition is parts per hundred: % (wlw) (mass of solutelmass of solution) x 100. For very dilute solutions, chemists will often use parts per thousand or parts per million (



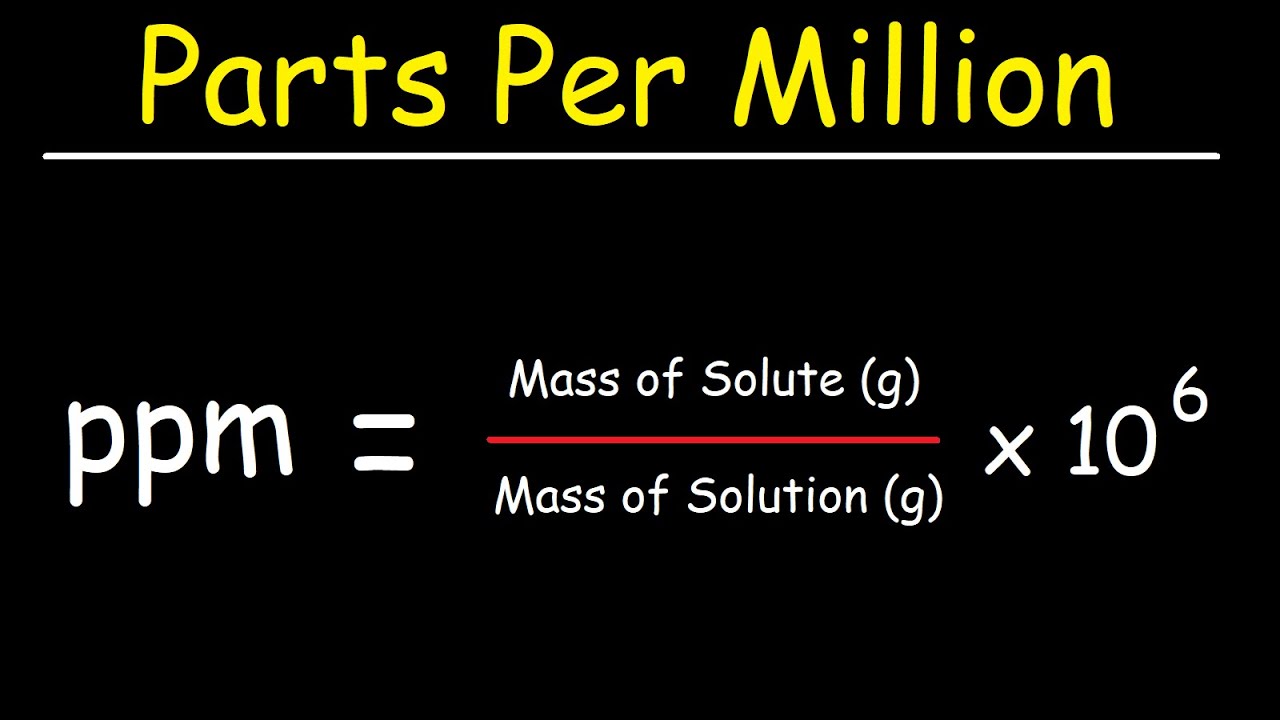





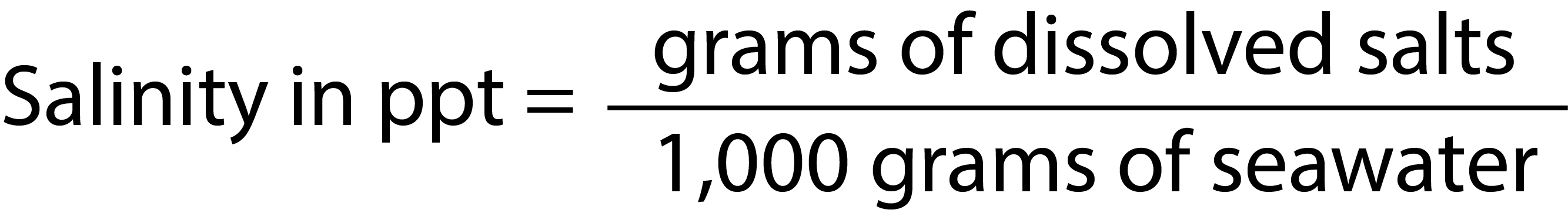






![Quantifying Precision | Calculating a Parts-per-Thousand [ppt] - YouTube Quantifying Precision | Calculating a Parts-per-Thousand [ppt] - YouTube](https://i.ytimg.com/vi/EyvTuj8ohQM/maxresdefault.jpg)